Definitive TNF Therapies Inc
The Definitive Oral
Anti-TNF Therapy
Developing macrocyclic anti-TNF therapeutics that combine antibody-like target engagement with the convenience and safety of a once-daily pill.
Request Investor DeckMarket Context
The Opportunity
Anti-TNF is one of the most proven markets in medicine, with more than 7.5 million patients treated annually and about $44 billion in yearly sales. Yet every approved anti-TNF still requires injection or infusion. The need for an oral anti-TNF remains unmet.
Oral drugs are usually small molecules, but TNF presents a broad, shallow receptor-binding surface spread across a large interface. Molecules small enough to be absorbed can engage only part of that surface, which has not been sufficient to block signaling. Antibodies can cover the surface effectively, but they do not survive the gut or cross the intestinal wall.
Macrocyclic peptide drugs occupy the space between small molecules and biologics. Some have been orally bioavailable for decades, beginning with cyclosporine in 1983, but for most of that history they were discovered by exception rather than by design. As the principles governing oral absorption for these molecules have become better understood, rationally designed macrocyclic peptides have begun advancing in the clinic. The modality is no longer a fortunate exception. It is an engineerable drug class.
The remaining challenge is access to the chemical space needed to engage a target like TNF while maintaining oral bioavailability. DTNF's programs are built on a proprietary enzymatic macrocyclization platform that produces molecules conventional chemistry and current macrocycle platforms cannot. These molecules are designed to be large enough to cover a target surface like TNF's while possessing the physicochemical properties necessary for oral absorption. They are synthesized compounds from an actively screened library, not hypothetical structures generated only in silico.
Pipeline
Dual Programs. Parallel Angles of Attack.
We run two programs against the same pathway through the same screening infrastructure. Program A replicates what Humira does in a pill. Program B goes further, blocking only the receptor that drives inflammation while leaving protective signaling intact.
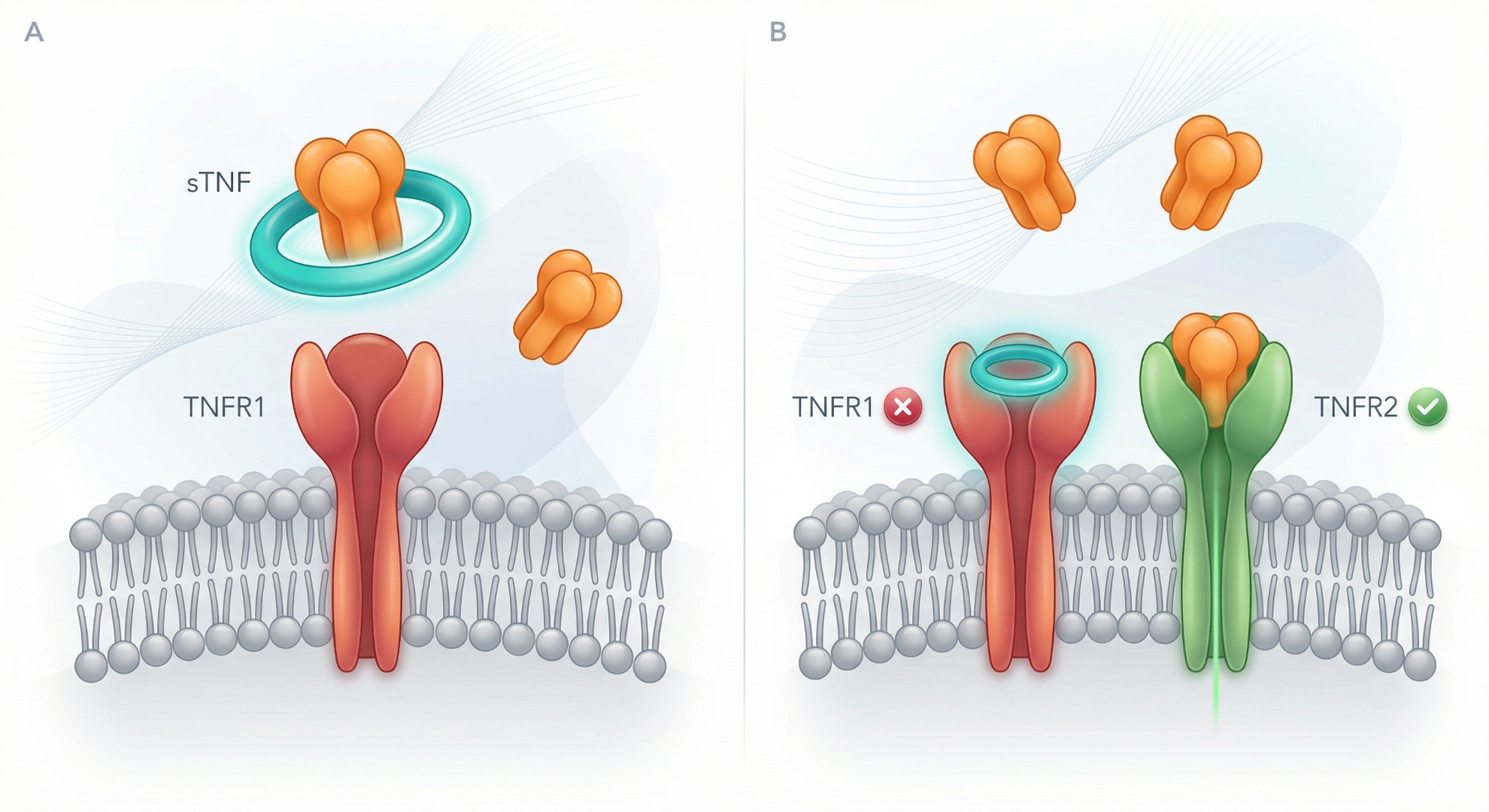
Program A: The Floor
Direct TNF Ligand Interception
Designed to replicate the mechanism of established anti-TNF biologics in an oral macrocyclic peptide format. Our goal is to eliminate injection burden and cold chain logistics, while reducing risks associated with anti-drug antibodies seen with biologics. Proven biology, new modality.
Program B: The Ceiling
Selective TNFR1 Blockade
Current anti-TNF biologics block TNF indiscriminately, suppressing both the inflammatory signaling mediated by TNFR1 and the protective signaling mediated by TNFR2. Program B targets TNFR1 alone. A viable TNFR1-selective oral agent would redefine who can take an anti-TNF and when: earlier lines, longer duration, broader populations.
How We Operate
Capital Efficiency by Design.
DTNF is an asset-centric company. We exist to advance an orally bioavailable anti-TNF asset through to clinical proof of concept.
We keep strategy, IP, and stage-gate decisions in-house. Everything else is executed by external partners selected for the exact capability each stage of development demands.
This is not a cost-saving measure. Drug development requires different expertise at each phase: chemistry at discovery, process development at CMC, regulatory affairs at IND, clinical operations in the clinic. Those needs rotate. Permanent infrastructure for all of them is structurally inefficient and creates incentives to keep programs alive longer than the data supports.
Our model eliminates that. More than 80% of capital goes directly into advancing the science with spending tied to scoped and defined milestones.
Our Team
From Discovery to Clinic.

Chadwick Davis, PhD
Founder & CEO
Drug development operator who converts early-stage discoveries into clinical assets. Employee #6 at Recursion Pharmaceuticals, where he joined pre-seed, helped build the phenomics platform, and led programs from hit identification through development candidate nomination across multiple therapeutic areas. He has built translational programs across neurodegeneration, inflammation, and rare disease, and has sat across the table from pharma partners in licensing and collaboration discussions.
He started DTNF to take one from platform to patient himself. TNF is one of the most validated verticals in medicine and after three decades, nobody has put it in a pill. DTNF is developing enzymatically engineered macrocyclic peptides to do it.
PhD in Human Genetics, University of Utah. Published in Nature, Neuron, Circulation, and the Journal of Immunology.

Karsten Eastman, PhD
Founding Scientific Advisor
Creator of the radical SAM maturase platform that enables DTNF's macrocyclic peptide chemistry. CEO of Sethera Therapeutics and the scientific mind behind the enzymatic cyclization approach underpinning both DTNF programs. Named 2025 University of Utah Founder of the Year. PhD in Chemistry, University of Utah. Published in PNAS, ACS Central Science, and JACS.
Our Network
Trusted Partners.
Every partner was selected for a specific reason: they have already delivered in the exact capability our programs require. Together, they form the integrated engine behind our design-make-test-analyze cycle, from proprietary macrocyclic chemistry through clinical execution and exit-ready legal strategy.

Sethera Therapeutics
Chemistry & Platform
Sethera's proprietary enzymatic macrocyclization platform gives DTNF access to polymacrocyclic peptide architectures that combine the target engagement surface required to neutralize TNF with the permeability profile required for an oral medicine. The platform was co-invented by our founding scientific advisor, who leads the team at Sethera that synthesizes every polymacrocyclic peptide across both of our programs. The people making our molecules are the people who built the chemistry.

Molecular Forecaster
Computational Chemistry (CADD)
Macrocyclic peptides sit in beyond-Rule-of-5 chemical space, where 3D flexibility and environment-dependent conformations break conventional small-molecule design tools. We run computational triage before synthesis, modeling binding modes, conformational ensembles, geometry, and permeability to rank candidates before they ever reach a bench. Our computational partner, Montreal-based Molecular Forecaster, built their platform for exactly this chemical space. For a seed-stage company on a defined timeline, this means our synthesis budget goes to the right molecules and we get there faster.

PI Health Sciences
Integrated CRDMO (Discovery through Manufacturing)
PI Health Sciences is our end-to-end discovery and development partner. A global CRDMO with facilities spanning R&D through cGMP manufacturing, PIHS designs and executes our preclinical campaigns from binding characterization through functional validation in disease-relevant systems. Their scientific advisory bench includes veterans of Bristol-Myers Squibb, Daiichi Sankyo, Agios, and other world-class pharma. Working alongside our chemistry and computational partners, the pharmaceutical leaders at PIHS close the design-make-test loop at the center of our virtual model and ensure we're ready for our CMC scale-up.

MedPace
Clinical Operations & Regulatory Execution
MedPace is a full-service CRO that built its reputation on emerging biotech companies. They are our clinical execution partner from IND planning through early proof of concept, selected for demonstrated depth in inflammatory disease and a proven model for working with lean virtual teams. MedPace delivers an integrated stack across regulatory support, protocol development, clinical monitoring, data management, biostatistics, medical writing, and pharmacovigilance. We engaged them at the seed stage so the relationship, infrastructure, and program readiness are in place before IND filing, not assembled after the fact.

Goodwin Procter
Legal
Goodwin is a nationally ranked life sciences law firm with over 230 dedicated lawyers advising biotech companies from formation through exit. In the past three years alone, Goodwin has represented Blueprint Medicines in its $9.5 billion acquisition by Sanofi, SpringWorks Therapeutics in its $3.9 billion sale to Merck, and Nimbus Therapeutics across the $1.2 billion sale of Nimbus Apollo to Gilead and the $4 billion sale of its TYK2 program to Takeda. Nimbus is the closest structural parallel to DTNF: a virtual, asset-centric company built around subsidiary programs designed for sequential strategic acquisition. We chose Goodwin because they have executed this playbook repeatedly, at the scale we are targeting.
Connect
Let's Talk Pipeline.
DTNF is raising $2.2M to advance the programs through lead optimization and begin IND-enabling studies.
Request our investor deck and data room access:
investors@definitivetnf.comFor all other inquiries:
info@definitivetnf.comSalt Lake City, Utah